Introducing Solutions (which, by the way, are not all aqueous!)...
|
Let's start with one of those fun comparisons where I tell you "all solutions are mixtures but not all mixtures are solutions." Knowing that solutions are mixtures, however, reminds you that these are systems created by physical changes, with one thing (which we'll call the "solute" gets dissolved in another thing (which we'll call the "solvent") when mixed together by a chemist (who we'll call "Bob"). So what makes solutions so special? They are homogenous meaning the particles of both the solvent and solute are evenly distributed throughout the mixture.
|
Water, one heck of a solvent!
Water-based or "aqueous" solutions also conduct electrical current. Pure water, no, but when you dissolve salt or other electrolytes into water then yes! The higher the concentration of ions or electrolytes in solution, the better the conductivity.
|
Making solutions
To make a solution in a chemistry lab: 1) Grab a volumetric flask calibrated to measure the specific amount of solution you want to make, 1L, 2L , etc., 2) calculate the mass (or volume) of solute you need to add to meet your desired concentration (molarity), 3) add a small amount of solvent and all of the solute to the flask, stir until solute dissolves, and finally 4) fill the flask to the calibration line with additional solvent.
|
Speaking of concentration... (aka "Molarity")
|
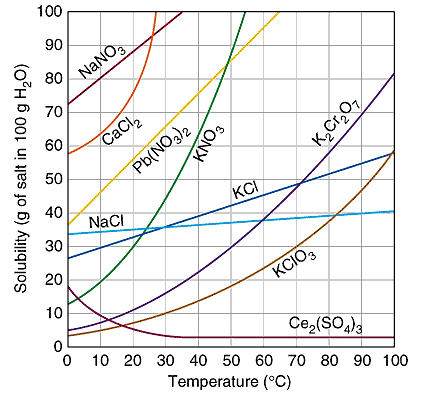
Solubility curves
|