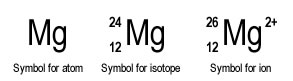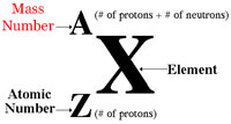What are Atoms?
|
Atoms are the smallest pieces of matter that have a unique chemical fingerprint. Atoms of an element all share the same basic chemical properties.
Some books say all atoms that make up the same element are all "identical," which is mostly true, but be careful! While all atoms of the same element do have an identical number of PROTONS, you will learn about ISOTOPES and IONS (which are also atoms) that have differing numbers of neutrons and electrons. What are the parts of an atom? These resources will help you understand the nature and parts of the atom:
How do you write the symbol for an atom? Well, we usually think of this as writing symbols for elements, but it's the same idea. Each element (and therefore its component atoms) has a unique symbol on the Periodic Table. Some instantly make sense, like O for oxygen or C for Carbon. Some are weird, like Pb for lead or Na for sodium. Remember many elements were named in Latin or Greek. The point is the symbol will either be a single upper case letter (H, K, C, N, O) or an uppercase and lowercase pair (Na, Mg, Cl, Xe, Cu). In two-letter symbols the second letter has to be written lower case: NA or CL is just plain wrong. How much do you need to know about atoms?
|
What are Isotopes and Why Do They Exist?
|
You can think of isotopes as atomic "siblings." They are part of the same family (same number of PROTONS) but they are unique in that each isotope has a different number of NEUTRONS in its nucleus.
The MASS NUMBER of any ion is the sum of its PROTONS and NEUTRONS. Istotopes have the same ATOMIC numbers (number of protons) but different MASS numbers (number of neutrons).
Why do we have isotopes? Isotopes are still a bit mysterious. I have never found a great, definitive explaintation of why they exists. Scientists agree neutrons help stabilize the nucleus of the atom. Think about it...a close packed group of protons (all with a + charge) would repel each other so neutrons act like little charge buffers. But why an element can be made up of so many different isotopes and why some elements have more stable isotopes than others? Still kind of a mystery. When we talk about radioactive decay you'll also learn that isotopes can break down and turn into other isotopes or even other elements! These resources will help aquaint you with isotopes:
How do you write the symbol for isotopes? You've already seen how to write the name of an isotope: (Element Name) - (Mass Number of Isotope) like Carbon-12, Carbon-13, and Carbon-14. To write the symbol for an isotope you just add a few things to the the basic symbol for an element. The UPPER number represents the MASS NUMBER, which is number of protons PLUS number of neutrons. This is unique for every isotope. Magnesium 24 has 12 neutrons, Magnesium-26 has 14 neutrons and Magnesium-30 has 18 neutrons. All Magnesium isotopes have 12 protons.
How much do you need to know about isotopes?
|
What are Ions and Why Do They Form?
|
Ions are atoms that have either lost (given away) or gained (accepted) electrons.
Why do we have ions? Every element dreams of looking like a Noble Gas. Ah, stability! It's the atomic equivalent of a restful vacation on a tropical island. To "look like" a Noble Gas, atoms need to have full outer orbitals (or as full as they can get). When we get to electron configuration you'll see just how that process happens, but for now just understand this:
Cool trick: The number of valence electrons for atoms/elements in Groups 1A-8A match the Group A number! Group 1A atoms all have 1 valence electron; Group 2A atoms all have 2 valence electrons; Group 8A atoms all have 8 valence electrons. How can you tell if an atom will give away or grab electrons? We'll study the Periodic Table in more detail soon, but for now think about it this way. Whether you give away or grab electrons you need to spend energy, and the "magic number" needed to fill your s- and p- orbitals is 8 (focus on the number 8 not the s-, p- thing).
Think about it - Why are anions (-) larger than cations (+)? Because a cation gives away valence electrons so it doesn't need to support a partially empty orbital. Once the electrons are gone - POOF - bye-bye orbital. The cation is SMALLER than the atom. On the flip side an anion grabs as many electrons as it need to fill that partially empty outer orbital. The orbital remains, except now it's happily full. BOTH ions end up looking like a Noble Gas (ahhhhhh....stability!) but they do it in opposite ways. When sodium (Na) gives away its valence electron it looks like Neon, the Noble Gas in the previous row on the Periodic Table. If chlorine grabs that extra electron it looks like Argon, the Noble Gas in the same row on the Periodic Table. These resources will help acquaint you with ions:
How do you write the symbol for ions? To write the symbol for an isotope you just add to the the basic symbol for an isotope, namely the charge.
How much do you need to know about ions?
|
What is "Atomic Number?"
|
the number of protons in your nucleus DEFINES you as an element. It's your unique fingerprint. No two elements on the Periodic Table have the same number of protons in their respective nuclei. It's that important!
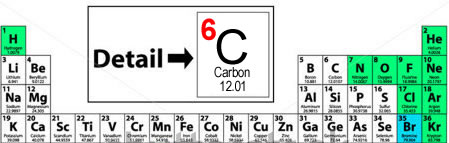
Atomic numbers - you'll learn soon enough - are also the basis for the underlying structure of the modern Periodic Table. Elements are arranged by increasing atomic number (that's one important little number!). Where do you find atomic number? On 99.9% of Periodic Tables (cuz you know there is a rebel PT out there), the atomic number is written above the symbol for the element. It can be centered above it or in the left or right corner. If you get confused, just look at the elements to the left and right. Since atomic numbers go up sequentially from 1-116 it's easy to pick it out in a crowd. Carbon's atomic number is 6. Boron's (B) is 5, Nitrogen's (N) is 7. These resources will help you understand the concept of atomic numbers:
How much do you need to know about atomic number?
|
What is "Mass Number" and How is it Different from "Atomic Mass?"
|
Okay it does seem like we have a lot of numbers with very similar names. Here's a simple summary, then we'll look at mass number and atomic mass in more detail.
Where do you find mass number?
Where do you find (average) atomic mass?
These resources will help you understand the concept of (average) atomic mass and show you how to do average atomic mass calculations:
How much do you need to know about mass number and atomic mass?
|
PNE (proton, neutron, electron) Calculations
|
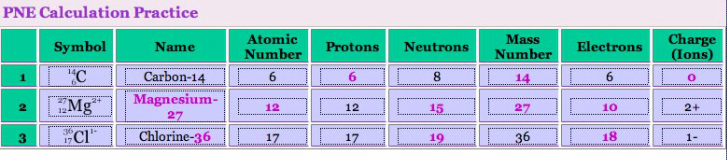
As redundant as it may seem let's look at these definitions one more time. It will help you when you start doing PNE calculations:
Knowing this you can figure out the "missing" pieces in the PNE puzzle. You should be able to figure out what's shown in pink below based on what's given in black. Review the table below then try the practice problems in your packet. Some helpful hints if you're still a bit confused:
These resources will also help you learn and practice PNE calculations:
|