Heat vs. Temperature
|
Once again chemistry gives us the chance to rethink the English language. For example, we tend to say things like "it's hot outside, it's 100 degrees (Fahrenheit, that is!)," but in reality "heat" and "temperature" are very different. In fact (technically) if two objects are at the same temperature, there is no "heat!" (Heat capacity, yes, heat, not so much). Confusing, no?
|
Endothermic vs. Exothermic
|
Heat (energy) always travels from hot to cold; the hotter object loses energy and the colder object gains energy until both reach a state of thermal equilibrium where their temperatures are the same.
|
Enthalpy and ΔH
|
Enthalpy is the amount of heat absorbed or released by a system at constant pressure. We are going to focus on enthalpy change or "ΔH". If the system absorbs energy then ΔH will have a positive value (e.g. "ΔH = +1200 kJ). If the system releases energy then ΔH will have a negative value (e.g. "ΔH = - 950 kJ). Here the (+) and (-) indicate the direction that energy is being transferred.
|
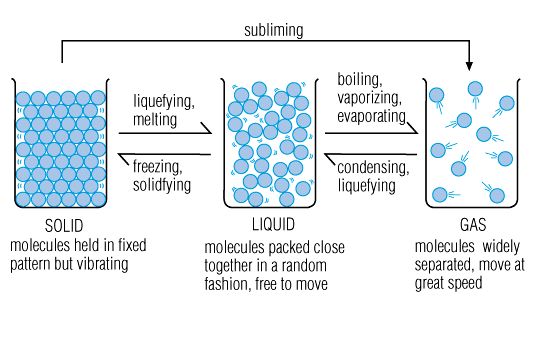
Phase Changes or Changes in State
|
You can also calculate the total ΔH of a series of endothermic or exothermic processes. Let's start with something familiar, heating a block of ice until it becomes steam. The water molecules never chemically change during this process, but the way they move does, and that's what allows phase changes or changes of state from solid to liquid to gas. Along the way the temperature (motion of the molecules) will also change as they absorb energy from the surroundings.
|
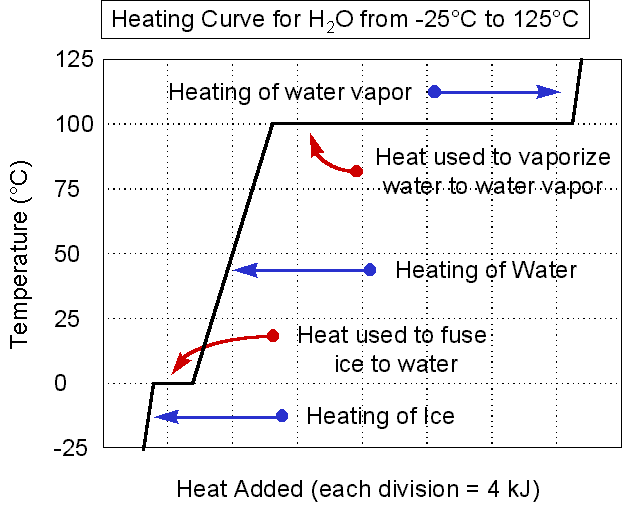
Heating Curves
|
|
Right about now you're probably wondering how it's possible that energy is constantly being absorbed by the system but the temperature isn't always changing. That's because at different points along the way the energy being absorbed is used to do different things! Here are the basics (we'll look at water, but keep in mind melting and boiling points vary for different substances):
|
Phase Diagrams
|
Calorimetry
|
A calorimeter is a device used to measure heat of reaction. It can be sophisticated and expensive or simple and cheap (the one we're using in lab is made of a Styrofoam coffee cup with a plastic lid. Judge for yourself!)
|