What is the Periodic Table?
|
|
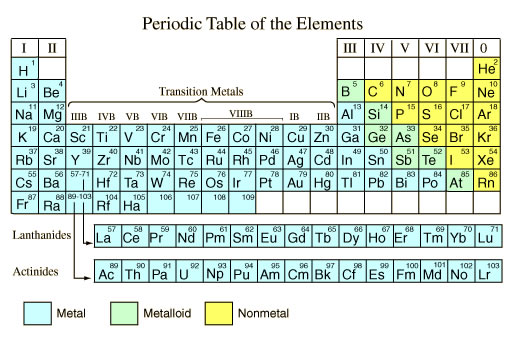
It's a structure for organizing chemical elements. The modern Periodic Table is arranged in order of atomic number.
Here are some great online periodic table resources! Before you read up on the history and logic of the structure just click in and look around the Periodic Table.
Do we really need a Periodic Table? Well, yes! We humans are great organizers: closets, sock drawers, play lists, tool boxes, athletic equipment. We sort, organize, and store things according to logical groupings because it makes things faster to find and easier to use. Imagine how long it would take you just to get dressed and out the door every day if all your stuff was just tossed around in random piles, or how long it would take you to find your favorite music if you had no way to create play lists or sort by song or artist. Time wasting mayhem. The structure of the modern Periodic Table follows the Periodic Law. Elements are arranged from 1 to (currently) 116 based on atomic number (number of protons in the nucleus, remember?), which means groups or families of elements exhibit similar, predictable, physical and chemical properties. You'll learn all about that in the "Trends" section. For now just trust me, if you are a chemist (and deep down there's a little chemist in all of us) you will grow to know and love your Periodic Table. These resources will help you understand the basics of the Periodic Table:
|
A Brief History of the Modern Periodic Table
|
Although Dmitri Mendeleev is often credited as the "father" of the modern periodic table (it would still be my Cash Cab answer choice if asked) there were actually many scientists who contributed. You need to now these few key players:
In a nutshell as more and more elements were discovered, compared and analyzed, the need for way in which to organize them became increasingly important. The evolution of the modern Periodic Table makes sense if you think about it. The very first Periodic Table, written by Antoine Lavoisier in the late 1700's, was basically a list of the then 33 elements grouped as metals or non-metals. In 1828 Jons Jakob Berzelius contributed the idea of atomic weights and added symbols. In 1829 John Doberiener introduced the idea of "triads" or groupings elements with similar properties. And so it went over the years with different scientists refining and adding to what is now a well organized tool that not only organizes existing elements but lets us predict the properties of new, as of yet undiscovered elements. You need to focus on these two:
For you history buffs, these links give you an expanded history of our little friend:
How much do you need to know about the history of the Periodic Table?
|
How is the Modern Periodic Table Organized?
|
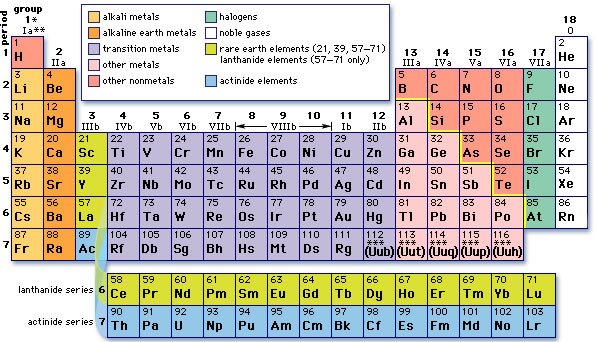
The common modern periodic table is organized by atomic number (thanks, Mosley!). From there we classify metals vs. non-metals (and metalloids) and group elements into Families like Alkali Metals, Halogens, and Noble Gases.
Groups vs. Periods
Metals vs. Non-Metals The first big grouping within the modern Periodic Table is whether an element is a metal, non-metal, or metalloid (semi-metal).
Groups/Families
Elements in the same Group or Family tend to have the same general characteristics. These are the Groups you need to know:
Here are some other links to help you get acquainted with all the Groups/Families:
How much do you need to know about the Periodic Table is Organized?
|
What Do We Mean by "Periodic Trends?"
|
Many, many physical and chemical properties 'trend' by Group and Family in the Periodic Table. We're going to cover a few of the biggies.
How much do you need to know about the Periodic Table trends?
|
Trends Around the Periodic Table
|
Atomic Radius:
Ionization Energy:
Electronegativity:
Chemical Reactivity:
METALS DOWN a Group: In METALS reactivity INCREASES as you go DOWN a Group because the farther down a Group of metals you go, the easier it is for electrons to be given or taken away, resulting in higher reactivity. ACROSS a Period: In METALS reactivity DECREASES as you go ACROSS a Period because though they still want to give away valence electrons they have more of them to get rid of, which requires more energy. Not as easy to blow off a little steam!
NON-METALS
UP a Group: In NON-METALS reactivity INCREASES as you go UP a Group because the higher up and to the right atoms are, the higher the electronegativity, resulting in a more vigorous exchange of electrons. Fluorine? A greedy, impatient beast when it comes to electron exchange manners. ACROSS a Period: In NON-METALS reactivity INCREASES as you go ACROSS a Period because (notice how trends repeat?) the closer you get to fulling your s- and p- orbitals the more motivated you are to do so. This links to the page specifically on TRENDS, but if you click around it also summarizes the other Periodic Table topics nicely. How much do you need to know about the Periodic Table trends?
|
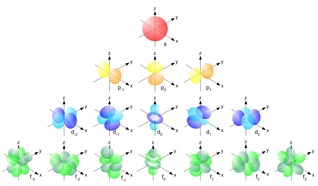
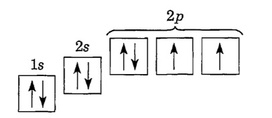
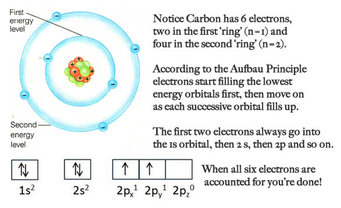
Honors Extension: What is Electron Configuration?
Writing Electron Configurations
|
Here are some other great resources on how to approach electron configuration:
Check out the videos below - Mr. Kent explains orbitals, electron configuration and the Periodic Table and writing electron configurations. 1-2-3 Understanding! (Thanks, Mr. Kent!)
How much do you need to know about Electron Configuration?
|