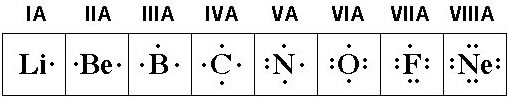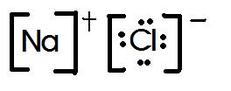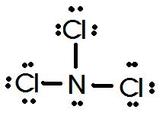Ionic vs. Covalent Bonds
|
Compounds form when elements bond chemically. There are two main types of chemical bonds: covalent and ionic (electrovalent). So what's the difference?
Use your Periodic Table to determine whether an element is a metal or a non-metal and how many valence electrons it has to help determine if it will bond by transferring (ionic/electrovalent) or sharing (covalent) valence electrons.
|
Lewis Dot Structures
|
Lewis Dot structures help you predict how atoms will bond because they show the valence electrons available for bonding. We're going to assume (for this class anyway) that bonding involves just the valence electrons. We already learned that atoms will ionize and transfer electrons (ionic bonds) or share electrons (covalent bonds) in order to reach the much coveted Noble Gas electron configuration. According to the Octet Rule* the magic number for such stability is "8" (2 electrons in an s orbital, 6 electrons in p orbitals).
Lewis Dot structures for Elements We also learned that for Main Group (Representative) elements, the number of valence electrons is the same as the Group A number; all Group 1A elements have one valence electron, all Group 2A elements have two valence electrons and so on all the way over to the Noble Gases in Group 8A with 8 valence electrons. The picture below shows the Lewis Dot structure for the elements in Period (row) 2. Notice how the number of valence electrons for each Group matches its Group A number! This should start your brain thinking... "yep," says your brain, "that explains why (in ionic bonding) metals give up electrons to gain a stable configuration while non-metals will gain electrons. If you're a metal it's easier to give up one electron than gain seven more; if you're a non-metal it's easier to gain one electron than give up seven." (Smart brain.) Lewis Dot structures can also help you predict how covalent bonds will form when electrons are shared between molecules.
Lewis Dot structures for Ions and Simple Compounds So what does a Lewis Dot diagram look like AFTER an element becomes an ION? What do Lewis Dot diagrams for compounds look like?
|