The Basics of Chemical Bonding
It's better to start by thinking of everything as the result of attractive forces - some between atoms in a single molecule and others between atoms in different molecules. Early on I told you that understanding how magnets work (like charges repel, opposites attract) will help you understand many underlying concepts in chemistry. Still true. The forces that bond atoms together are the same basic electrostatic attractions that make opposite poles of a magnet attract or that keep electrons attracted to the protons in the nucleus.
These resources will help you understand the basics of bonding:
The Octet Rule* When we studied electron configuration you learned that s orbitals hold 2 electrons, p orbitals hold up to 6 electrons, d orbitals hold up to 10 electrons, and f orbitals hold up to 14 electrons. So why all the emphasis on 8 ("octet")? Because 8 valence electrons between the s (2 e-) and p (6 e-) orbitals is the configuration of the Noble Gases! Notice every Noble Gas ends in an s2p6 configuration.
|
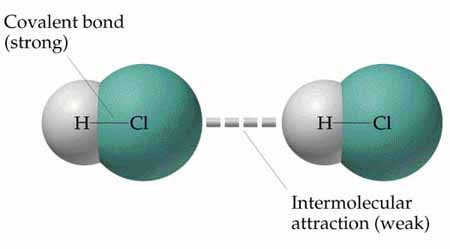
So What's an "Intermolecular Force?"
|
Let's start with a sports analogy. In "intermural" sports your team plays teams from other schools while in "intramural" sports teams within your school play each other. Something similar goes on in chemistry. Intermolecular forces (IMFs) attract a group of molecules to each other, like a league roster holds teams from different schools together in a conference, while intramolecular forces (bonds) hold atoms together within the same compound.
The bonds that hold two hydrogen atoms to one oxygen atom in a water molecule are "intramolecular" forces while the attractive forces that make water molecules stick to other water molecules are "intermolecular" forces. Click the "IMFs Only" tab to learn more about intermolecular forces. |