Calculating Atomic and Molar Mass
|
At the start of the year we talked about "average atomic mass," the weighted average of all of an element's isotopes. You find it on the Periodic Table. In the Mol (mole) unit we are going to revisit and apply that average atomic mass in a new way - to calculate ATOMIC and MOLAR MASS.
|
So What is a "Mol" (or a "Mole?") Exactly?
|
By definition, one mole is the number of atoms in 12 g of Carbon-12.
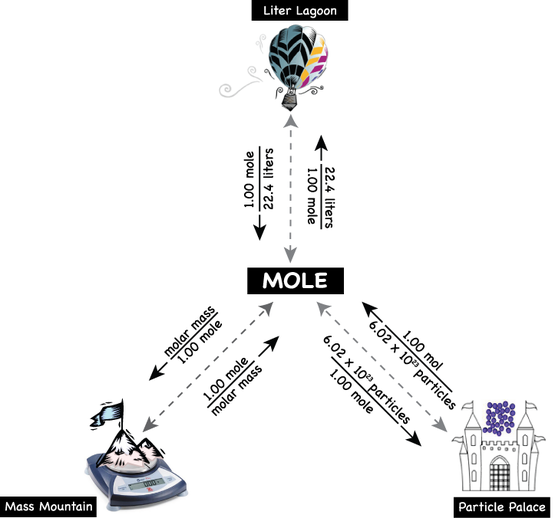
In chemistry we need to measure huge numbers of very, very tiny things. Using familiar units like "dozens" just won't cut it, so we use the "mol" (mole). As units go the mol is very versatile because it can be used in three ways: 1. A mol can be used as a COUNTING number, just like:
2. A mol can be used to measure VOLUME of a gas at STP:
3. A mol can be used to express the MASS of a group of atoms, molecules, or compounds
|
How "Big" is a Mol? Let's Explore Avogadro's Number!
|
The number of particles in one mole, 6.02 x 10^23, is known as Avogadro’s Number or Avogadro’s Constant. The constant was named after Lorenzo Romano Amedeo Carlo Avogadro di Quaregua e di Cerreto (1776-1856) whose ideas were crucial to the early development in Chemistry. Ironically Avogadro never knew his own number! Josef Loschmidt named it “Avogardo’s Number” many years later.
|
So the Mol Provides a Series of Conversion Factors. How Do We Use Them?
|
First let's make sure we understand WHY we need to do mol (mole) conversions! When we balance chemical compounds using subscripts and balance chemical equations using coefficients we are using MOL (mole) ratios. Thing is, the electronic balances or other measuring devices we use in lab can't give us a readout in "mols" (moles). We need to use conversion factors!
As you can see below, all conversion factors are anchored on the mol (mole). You can convert directly back and forth between moles and grams, moles and liters, and moles and particles. These are ONE-STEP conversion problems. You can also convert between liters and grams, atoms and liters, or atoms and grams indirectly using the mole as a "bridge." These are TWO-STEP conversion problems. So How Can You Tell Which Type of Mol (Mole) Problem You Have?
|