The Gecko Factor - The "Sticky" Chemistry of Intermolecular Forces
|
Geckos can "stick" to almost any surface! Read why here!
See how scientists are making gecko inspired adhesives (science in action!). |
I'll be honest with you - learning about intermolecular forces can feel a bit like trying to hit a fast-moving target! For one, as a community chemists are a bit inconsistent about terminology, and for another we keep redefining what "attractions" and even "bonds" are. This is the way I learned IMFs, so this is the way I'm going to teach them to you...
Intermolecular forces (IMFs) are different than bonds in two major ways:
All IMFs are collectively classified as "van der Waal's" forces. In my mind the easiest way to look the individual IMFs is to first divide them in two broad categories - "Temporary" and "Somewhat Permanent" (you will never see those terms on another website, but trust me...there is a method to my madness!).
So let's dig deeper into each one...
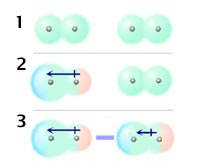
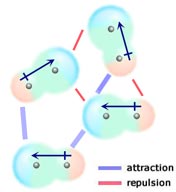
Dispersion Forces (Also called "London forces," after Fritz London who first proposed how they might work). We know electrons are always moving, so at any one instant they might find themselves bunched up at one end of a molecule, making that end partially negative and the other end partially positive. Two seconds later all the electrons can shift again and the temporary polarity reverses! This constant random, rapid motion of electrons makes it possible for even non-polar molecules to be attracted to each other. Temporary poles are always forming, and these temporary poles can even induce or create other temporary dipoles. If a non-polar molecule comes near a polar molecule the field from the polar molecule can induce a dipole moment in a nearby non-polar molecule. Spread this phenomenon across millions of molecules and you can see why whole lattice structures can form between non-polar molecules.
Dipole-Dipole Interactions
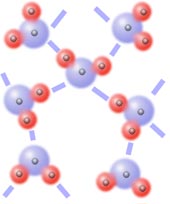
When polar molecules line up so the positive end of one molecule is near the negative end of another, they are attracted to each other by dipole-dipole interactions. Dipole-dipole interactions are effective only when polar molecules are very close together, like in liquids or solids (not gases). They are also generally weaker than ion-dipole forces.
Hydrogen "Bonds"
Okay, I'll just get this over with... I personally hate the term hydrogen "bond" because I think it's misleading. When true bonds break you get a chemical reaction; when hydrogen bonds are overcome you get a change in phase. So here's the deal - hydrogen "bonds" are just super-strong dipole-dipole interactions which form between a limited number of elements bonded in a specific sequence. One molecule must contain a H bonded to either N, O, or F and the other molecule must contain either N, O, or F.
Ion-Dipole Interactions
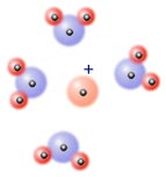
Remember "like dissolves like?" When an ionic substance dissolves in a polar solvent, most of the solvent molecules line up with the oppositely charged end of the solvent molecule near an ion. This attraction between the ions and the solvent molecules can win out over the attraction of the ions to each other, allowing the substance to stray in solution. Ion - dipole forces explain why salts and other polar solutes dissolve in water.
|