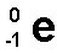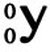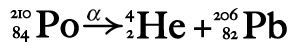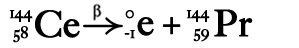What is Nuclear Chemistry?
|
Nuclear chemistry is the branch of chemistry that studies reactions that happen within an atom's nucleus.
We're only going to touch on a few of the basics. We'll start by defining radiation then look at how some elements undergo radioactive decay. For dessert we'll learn about and practice how to calculate half-life problems. Are all elements radioactive? Nope. Most of the 88 or so naturally occurring elements have pretty stable nuclei since neutrons keep the protons from repelling or pushing each other apart. Once you get past atomic number 82, however, all elements have radioactive isotopes. These isotopes will break apart or "decay," turning from one element into a different one - over and over again - until the nucleus is relatively stable. (By the way, there are a few lightweight isotopes like Carbon-14 that are radioactive, too. It's just more common among the bigger ones). The process of changing an unstable nucleus into a stable one is called "radioactive decay." You'll learn the details about how this works under "Types of Decay", but basically elements give off particles and/or rays that change the ratio of protons and neutrons in the nucleus. These resources will help you understand the basics of nuclear chemistry: How much do you need to know about nuclear chemistry?
|
What is Nuclear Radiation?
|
Nuclear radiation is the process during which the nucleus of an unstable atom spontaneously breaks down in an attempt to form a more stable nucleus.
You already know something about radiation in general. Sunshine is familiar; it gives us light, heat and an occasional nasty sunburn. Sunlight includes wavelengths from long-wave infra-red to short-wavelength ultraviolet. You may also remember something about high-energy (ionizing) radiation which can cause damage to matter, particularly living tissue. We're all exposed to low doses from space, the air, and even from the Earth itself. High doses of this ionizing radiation are dangerous so we have to be careful about exposure. That's why doctors and dentists give us those nice lead vests and leave the room before taking X-rays. We are also routinely exposed to low levels of naturally occurring background radiation. In the next section you'll learn more about alpha, beta and gamma raditaion, which is the main source of exposure for most people. Luckily there is no evidence it will hurt us at low levels. Where does this background radiation come from? It occurs naturally in our environment, coming from naturally radioactive materials in the ground and from cosmic rays. In addition to medical and dental X-rays, medical professionals use radiation to diagnose and treat a variety of diseases. On the dark side, radiation can kill! In fact, it's been used as a murder weapon: http://news.bbc.co.uk/2/hi/6190144.stm |
Types of Radioactive Decay
|
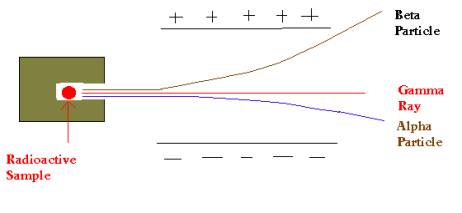
There are three main types of radioactive decay: alpha, beta, and gamma.
Let's pause here a minute to define "decay." When an element decays the parent element's nucleus changes - it will actually decay to turn into a different daughter element altogether! How is this possible? Because during radioactive decay the number of protons in the nucleus can change (I know, right?).
Alpha Radiation/Decay During Alpha decay an atom spits out two protons and two neutrons from its nucleus. This little bundle is called an "alpha particle."
Beta Radiation/Decay Remember we said a neutron is a proton with an electron attached? In beta decay a neutron sends its electron packing, literally ejecting it from the nucleus at high speed. The result? That neutron turns into a proton!
Gamma Radiation/Decay Gamma rays (remember that term from when we studied the EMS?) is electromagnetic radiation similar to light. Gamma decay does not change the mass or charge of the atom from which it originates. Gamma is often emitted along with alpha or beta particle ejection.
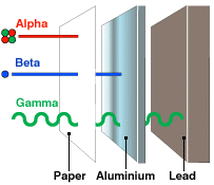
Comparison of Alpha, Beta and Gamma Radiation/Decay |
How Do You Write Equations for Alpha Decay?
|
Fair warning - these reactions look more complicated than they are! If you can subtract 4, 2, 1 or 0 from whole numbers you can write basic nuclear equations. Remember when an element spits out an alpha particle it loses two protons (changing the atomic number) and two neutrons (changing the isotope's mass number). Your job is to make sure all the mass numbers (top) and atomic numbers (bottom) add up.
Let's start with a non-chemistry example to prove this is a piece of cake (mmmm, cake!): 210 = 4 + ? Not so bad, right? The ? = 206 In a nuclear equation for alpha decay you'd be half done! This is how you balance the top part of the equation. So let's finish... 84 = 2 + ? Again, easy. The ? = 82. In solving a nuclear equation you're approaching the finish line! The 82 is the atomic number of the daughter element, so find element 82 in your trusty periodic table and you are done. |
How Do You Write Equations for Beta Decay?
|
Same idea and even easier since the mass number doesn't even change during alpha decay.
Remember when an element spits out a beta particle all it loses is one electron. It still changes the atomic number because what used to be a neutron is now an extra proton. Your job here is to make sure all the atomic numbers (bottom) add up. Once again we'll start with a non-chemistry example to prove this is easy cheesy (mmmm, cheese!): 144 = 0 + ? There's gotta be a catch, right? Too easy? Nope. The ? = 144. Remember the mass number doesn't change during beta decay. 50 = -1 + ? The only trick here is remembering you're working with negative numbers, so the daughter will have a HIGHER atomic number than the parent (extra proton, remember?). How much do you need to know about nuclear decay?
|
What is Nuclear Half-Life?
|
Nuclear half life is the time it takes one half of the atoms in a current sample to decay.
Sample size doesn't matter - whether you have one gram or one ton of something, half of the sample will decay during the fist half life. The sample will continue to shrink by half during each successive half-life until the atom becomes stable. Half-lives vary from less than a second to billions of years! Keep in mind that any sample or measurable size will contain billions of atoms, and there is no way to predict which specific atoms will decay next. You just know one half of them will decay during each half life. |
Solving Half-life Problems by Reasoning it Through
|
Even if you know nothing about using logarithms you can reason through basic half-life problems. Here's a simple example:
How much of a 20-gram sample of Sodium-24 will remain after 45 hours given that the half-life of Sodium-24 is 15 hours? Think about it:
It works in reverse, too! Just slow down and think about it. The half-life of Cobalt-60 has a half-life of 5.3 years. You find a 20.0 g sample that has been decaying for 21.2 years. Based on this information calculate the size of the original sample. Think about it:
If you are a ready to look at logarithms, take a look at the math way to approach half-lives! How much do you need to know about nuclear decay?
|